PGT之使用並無法明顯提升一般病患之累積懷孕率
PGT之使用並無法明顯提升一般病患之累積懷孕率
Live Birth with or without Preimplantation Genetic Testing for Aneuploidy
Published November 24, 2021
N Engl J Med 2021;385:2047-2058
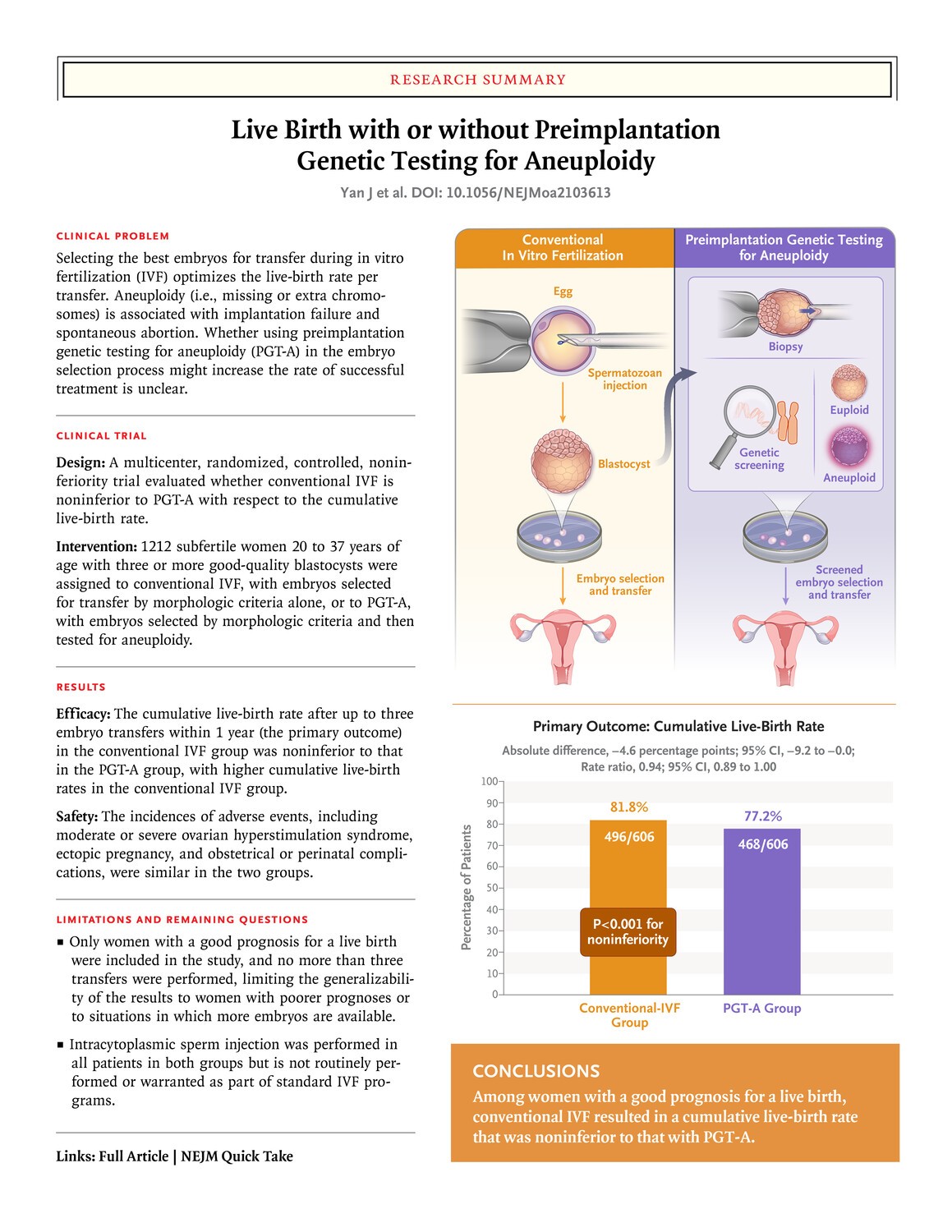
Embryo selection with preimplantation genetic testing for aneuploidy (PGT-A) may improve pregnancy outcomes after initial embryo transfer. However, it remains uncertain whether PGT-A improves the cumulative live-birth rate as compared with conventional in vitro fertilization (IVF).
METHODS
In this multicenter, randomized, controlled trial, we randomly assigned subfertile women with three or more good-quality blastocysts to undergo either PGT-A or conventional IVF; all the women were between 20 and 37 years of age. Three blastocysts were screened by next-generation sequencing in the PGT-A group or were chosen by morphologic criteria in the conventional-IVF group and then were successively transferred one by one. The primary outcome was the cumulative live-birth rate after up to three embryo-transfer procedures within 1 year after randomization. We hypothesized that the use of PGT-A would result in a cumulative live-birth rate that was no more than 7 percentage points higher than the rate after conventional IVF, which would constitute the noninferiority margin for conventional IVF as compared with PGT-A.
Download a PDF of the Research Summary.
RESULTS
A total of 1212 patients underwent randomization, and 606 were assigned to each trial group. Live births occurred in 468 women (77.2%) in the PGT-A group and in 496 (81.8%) in the conventional-IVF group (absolute difference, −4.6 percentage points; 95% confidence interval [CI], −9.2 to −0.0; P<0.001). The cumulative frequency of clinical pregnancy loss was 8.7% and 12.6%, respectively (absolute difference, −3.9 percentage points; 95% CI, −7.5 to −0.2). The incidences of obstetrical or neonatal complications and other adverse events were similar in the two groups.
CONCLUSIONS
Among women with three or more good-quality blastocysts, conventional IVF resulted in a cumulative live-birth rate that was noninferior to the rate with PGT-A. (Funded by the National Natural Science Foundation of China and others; ClinicalTrials.gov number, NCT03118141.)
N Engl J Med
. 2021 Nov 25;385(22):2047-2058.